Pan-screening detection: Less Complexity. More Confidence
Gastrointestinal infection is a major cause of morbidity in developed countries, and mortality in developing countries, causing the death of about 2 million children under 5 each year1. Gastroenteritis can be caused by a wide range of infectious agents causing similar signs and symptoms.
Genetic Signatures' advanced 3base® technology is transforming gastrointestinal diagnostics - delivering multiplex real-time PCR testing for 24 clinicallly relevant bacterial, viral, and parasitic pathogens in a single, automated workflow. The EasyScreen™ Pan-Enteric Detection Kit eliminates complex testing algorithms, reducing operational risk, hands-on time, and training requirements. Scalable to meet medium and higher throughput requirements, it empowers clinical laboratories to work faster and more effectively - without compromising on quality.
Beyond the laboratory
The benefits go beyond the lab. Faster, more comprehensive results support better clinical decisions, stronger antimicrobial stewardship, and quicker infection control responses - helping reduce unnecessary admissions, minimise ward closures, and shorten patient stays.
Contact us for more information on implementing this diagnostic solution in your laboratory.
Learn more about our gastrointestinal solution below.
1World Health Organization Children’s environmental health. Available at http://www.who.int/ceh/en/.
Syndromic Testing
Putting Patient Health First
Syndromic testing puts the patient first. Designing diagnostics around a specific set of symptoms to deliver more targeted, clinically meaningful results. This patient-centred approach streamlines the diagnostic workflow, reduces unnecessary testing, and supports faster, better-informed patient management.
✓ Testing all patients for the majority of clinically relevant pathogens
✓ Same day results
✓ Improved patient management and earlier treatment intervention
✓ Improved infection control
✓ Increased diagnostic yield
✓ Simplified processes and algorithms
✓ Reduced staff burden
✓ Reduced risk
✓ Epidemiological tracking
Conventional testing paradigm
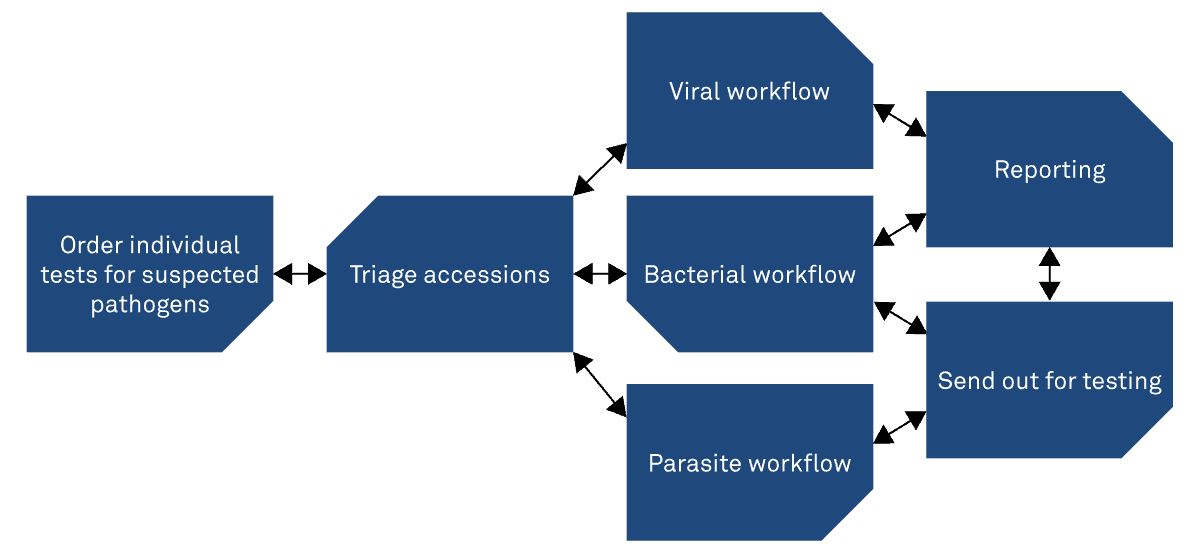
Syndromic testing

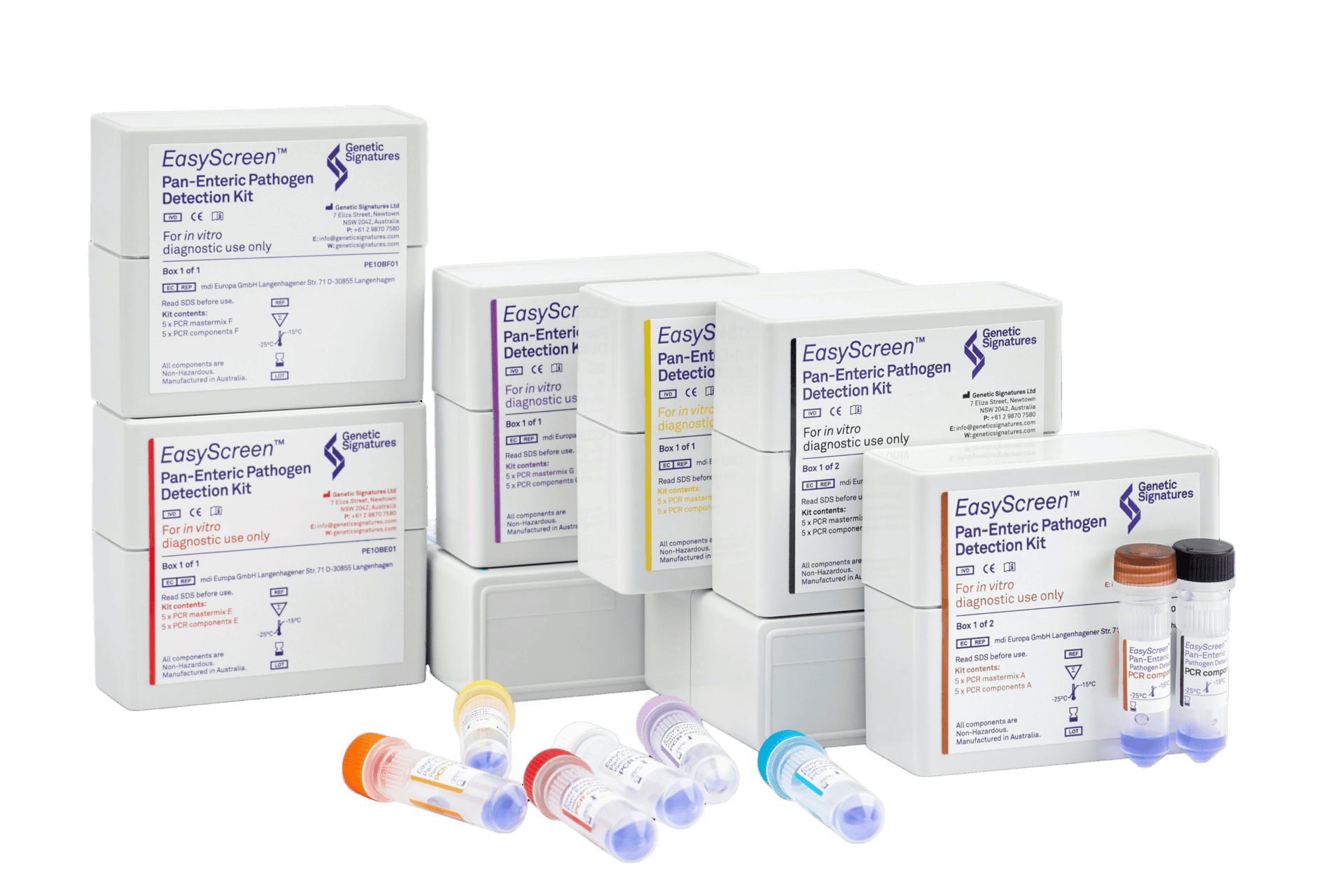
24 Targets.
Up to 42 samples per run.
1 Test.
Comprehensive screening of 24 clinically relevant targets across bacteria, viruses, parasites, and shiga toxins with a convenient, automated workflow.
EasyScreen™ Pan-Enteric Pathogen Detection Kit
REF PE001
Bacteria
- Aeromonas spp.
- Campylobacter spp.
- Clostridioides difficile (TcdA/B)
- E. coli - eaeA
- E. coli Labile Toxin (LT)
- E. coli Stable Toxin (ST)
- E. coli 0157:H7
- Salmonella spp.
- Shigella spp. / Enteroinvasive E. coli (EIEC)
- Vibrio cholerae
- Vibrio spp.
- Yersinia spp.*
* excl. Y. pestis and Y. pseudotuberculosis
Viruses
- Adenovirus 40/41
- Astrovirus
- Enterovirus
- Norovirus GI
- Norovirus GII
- Rotavirus A
- Sapovirus
Parasites
- Cryptosporidium spp.
- Entamoeba histolytica
- Giardia duodenalis
Shiga toxin
- Shiga toxin stx-1
- Shiga toxin stx-2
Benefits

Laboratory Benefits
• Quicker TATs
• Overall lab efficiency
• Fewer staff needed
• Timely results
• Faster clinical decisions
• Identify co-infections
• Supports antimicrobial stewardship

Hospital/Trust Benefits
• Infection control
• Improved patient & bed management
• Outbreak detection & surveillance
• Improved epidemiological tracking & health policy planning
• Overall cost saving
• Identify co-infections
• Supports antimicrobial stewardship

Patient Benefits
• Early diagnosis for earlier intervention
• Fewer hospital visits
• Shorter hospital stays
• Improved patient wellbeing
• Prevent outbreaks
• Protect staff
Automated Workflow
Our simplified, intuitive workflow combines sample processing (nucleic acid purification) and PCR setup into a single step on the GS1 automated platform.
All times are approximate.
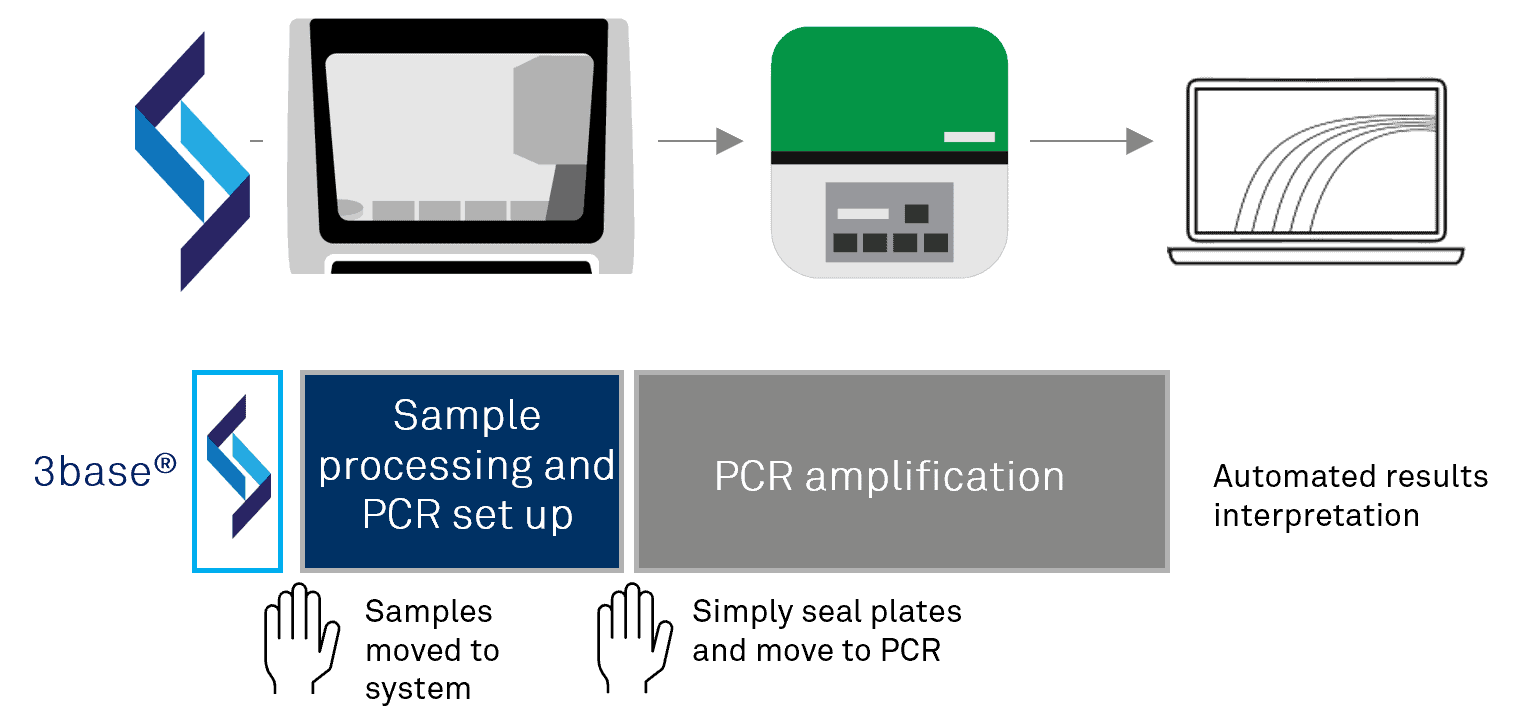
- Up to 42 samples tested simultaneously
- Convenient, automated workflow
- Parallel processing reduces sample-to-result time
- AI-driven results calling
- Automatic release of results to LIS
- Traceable reagent batches and expirations via barcode scanning
- Supports diverse stool sample types
Automated results interpretation software
GS-Analysis enables fast, reliable interpretation of results, whilst providing accurate and visible traceability of management processes.

Infection Control & Risk Reduction
Ward closures are most commonly driven by Norovirus and Clostridioides difficile, with pathogens such as Shigella and Cryptosporidium adding further transmission risks that demand heightened contact precautions and intensive environmental cleaning.
Comprehensive syndromic testing changes this picture. By enabling faster, targeted treatment and reducing length of stay, it eases the clinical and economic burden on both patients and hospitals — keeping wards open and care uninterrupted.
Estimated Cost of A Single C. difficile Ward Closure (UK)
|
|
Cost Category |
Typical Range | |
|
|
Lost bed-days |
£60,000 - £130,000 | |
|
|
Cancelled electives |
£30,000 - £50,000 | |
|
|
Deep cleaning |
£8,000 - £15,000 | |
|
|
IPC staffing & escalation |
£10,000 - £25,000 | |
|
|
Prolonged length of stay (subset of pts) |
£20,000 - £60,000 | |
|
|
Governance & reporting |
£5,000 - £15,000 | |
|
|
Estimated £ / ward event |
£130,000 - £300,000 |
Case Study
Real-world results speak for themselves - hear from Blackpool Teaching Hospitals about the benefit of employing Genetic Signatures's GI solution
A UK general hospital replaced a fragmented workflow of culture, ELISA, and multiple PCR methods with the EasyScreen™ Pan-Enteric assay across all faecal samples - with transformative results.
Turnaround times fell from 2.74 days to just 0.64 days, diagnostic yield tripled, and comprehensive results available within 5 hours enabled faster admission decisions, targeted antibiotic use, and earlier infection control.
Most strikingly, the hospital recorded a 23% decline in Clostridioides difficile infections - even as national cases rose by 33% over the same period, demonstrating the significant clinical and operational impact of comprehensive molecular testing.

Explore Workflow

View Webinar


